For US Healthcare Professionals
CLINICAL TRIAL
In the UNIVERSE clinical trial The first and only randomized study of a direct oral anticoagulant (DOAC) conducted to evaluate thromboprophylaxis in children post Fontan3
Objective
Randomized, multicenter, open-label, active-controlled, 2-part, phase 3 study to examine the use of a novel, oral suspension formulation of XARELTO® in children 2-8 years old with single ventricle physiology who had the Fontan procedure within 4 months before enrollment 2,3
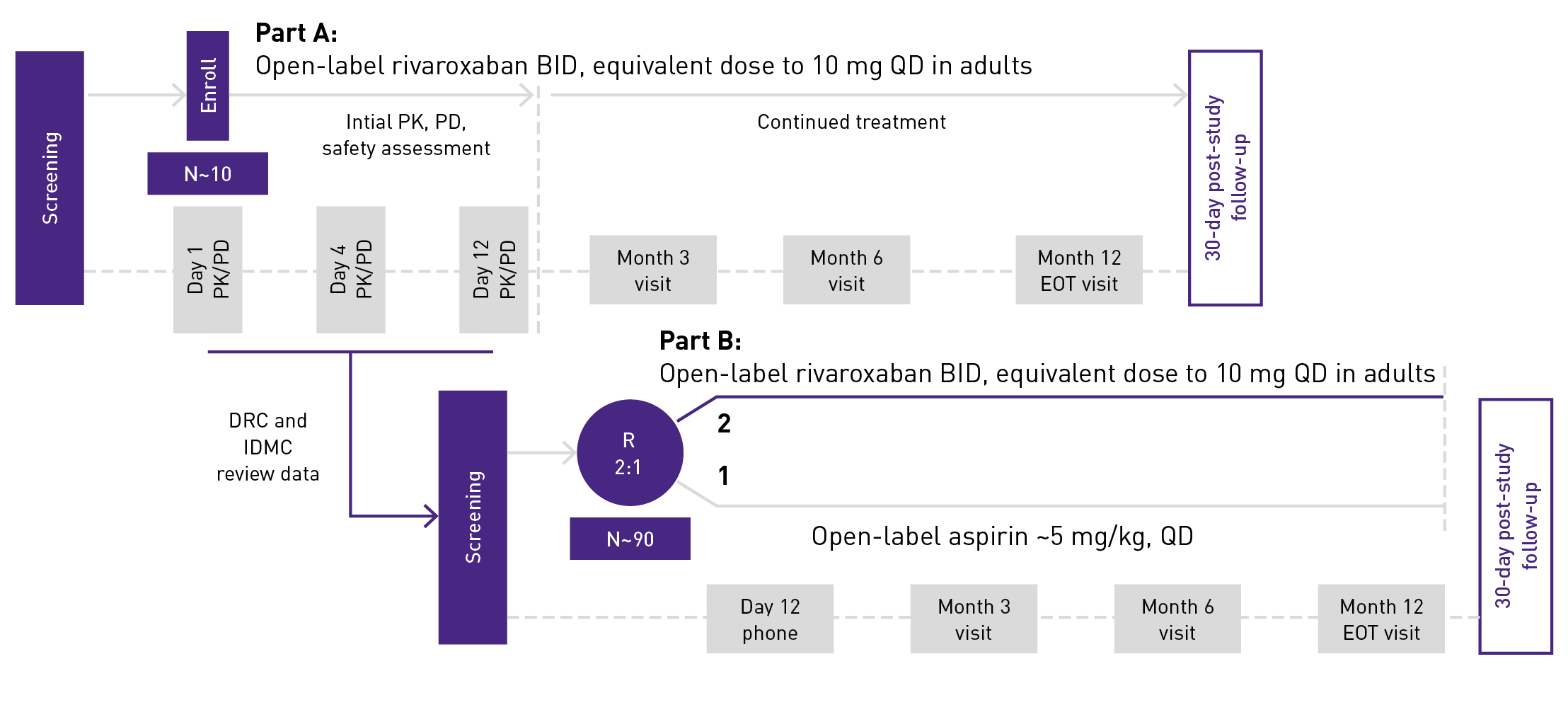
The UNIVERSE clinical trial comprised 2 parts2:
- Part A assessed the PK and PD of XARELTO® in young children to determine a dose that would approximate the drug exposure achieved in adults with XARELTO® 10 mg QD
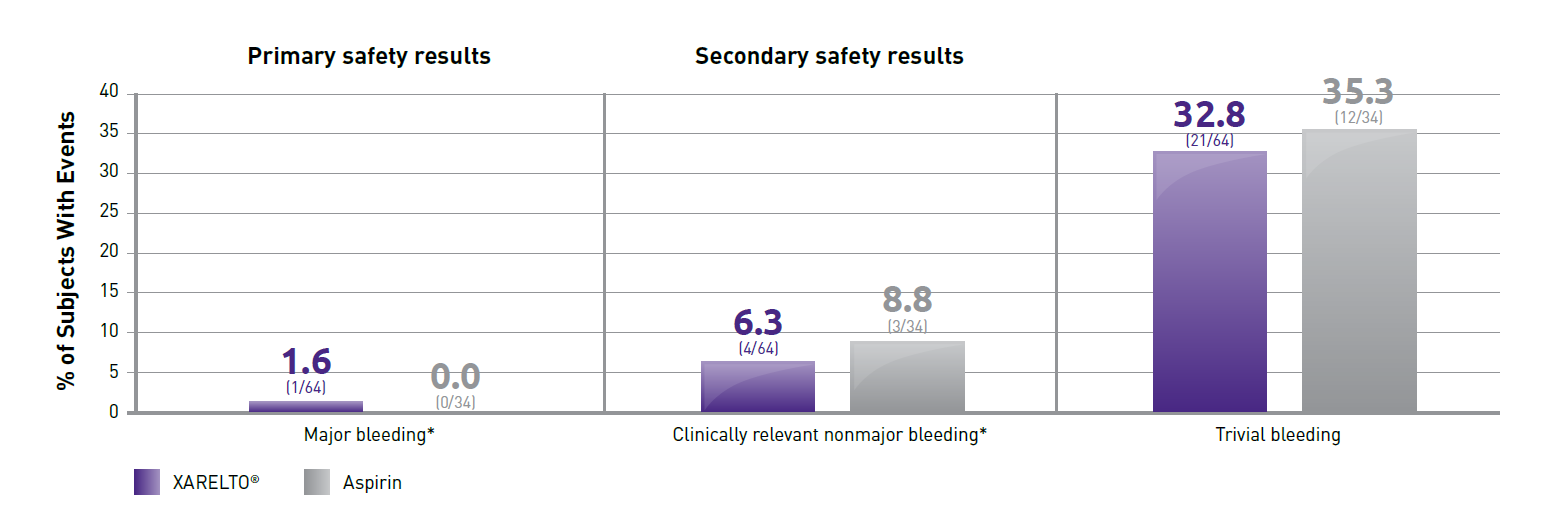
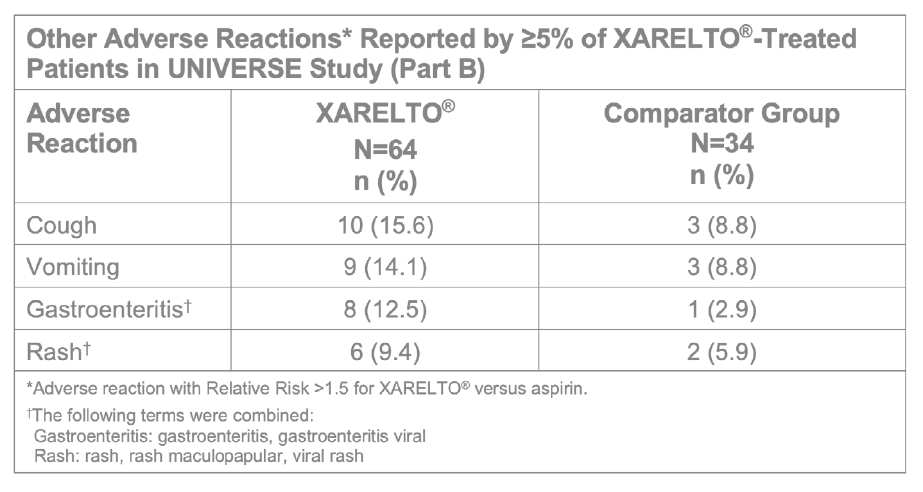
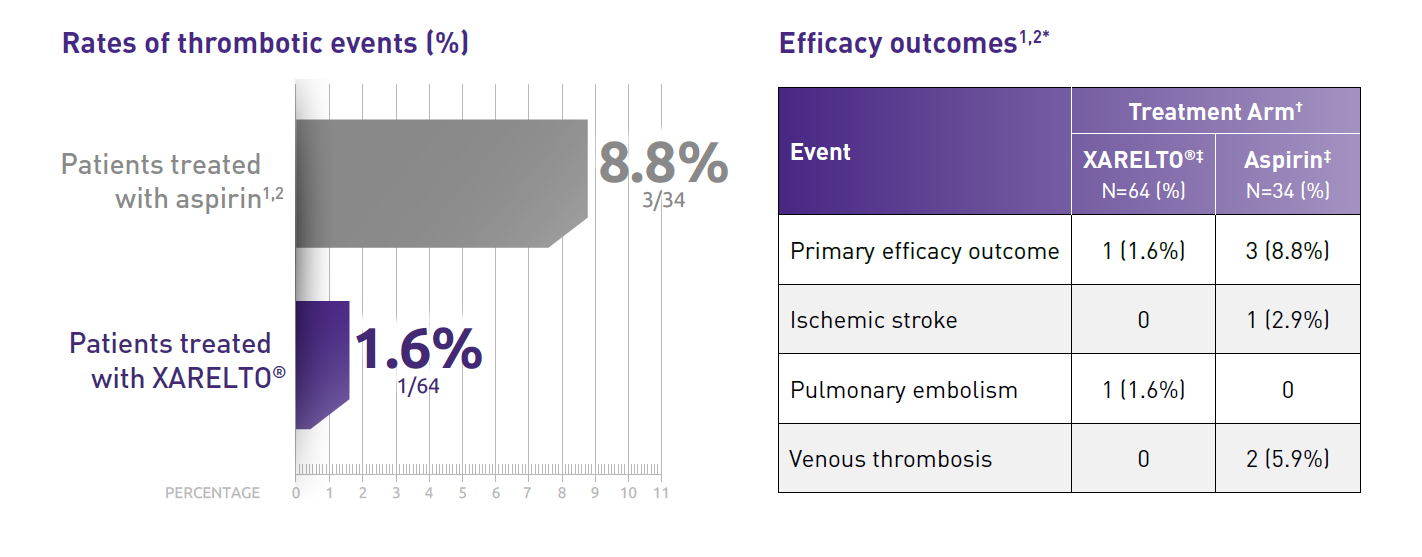
- Part B evaluated the safety and efficacy of XARELTO® vs aspirin for thromboprophylaxis post-Fontan procedure for 12 months
Not powered to test formal hypotheses for efficacy and safety due to the limited availability of the study population and the expected low event rates.2,3
BID = twice daily; DRC = Data Review Committee; EOT = end of treatment; IDMC = independent data monitoring committee; PD = pharmacodynamics; PK = pharmacokinetics; QD = every day; R = randomization.
UNIVERSE clinical trial study design2,3
STUDY POPULATION
- Children aged 2-8 years with single ventricle physiology, post-Fontan procedure (within 4 months prior to study enrollment)
- This was a VTE-prevention study; patients with thrombosis requiring treatment were excluded
-
Main exclusion criteria:
- Evidence of thrombosis
- History of GI disease or surgery associated with impaired absorption
- Active bleeding or high risk of bleeding contraindicating antiplatelet or anticoagulation therapy, including history of intracranial hemorrhage, or contraindication to aspirin or rivaroxaban
- Chronic use of NSAIDs
- Platelet count <50 x 109/L at screening
- Estimated eGFR <30 mL/min/1.73m2
TREATMENT GROUPS
- Subjects were randomized (2:1) to XARELTO® or antiplatelet therapy with aspirin (N=112)
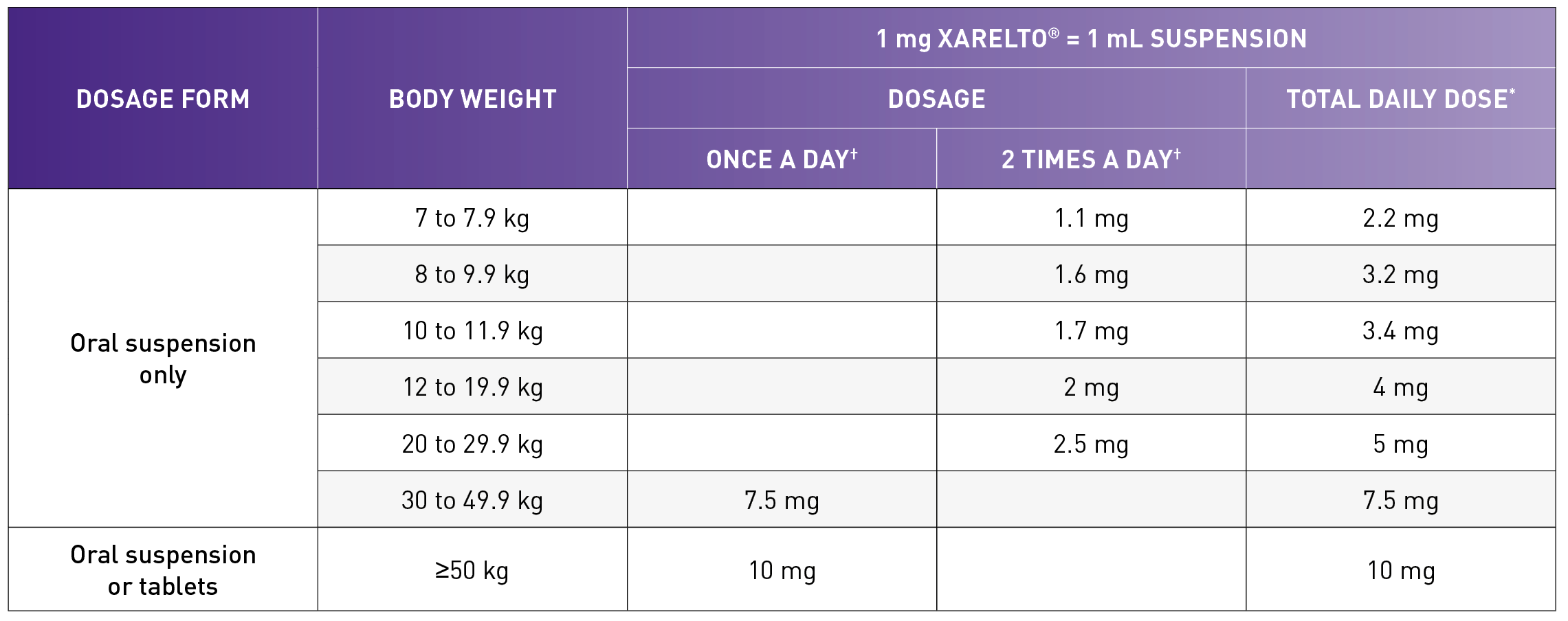
- XARELTO® was administered BID as a suspension; weight-adjusted to achieve drug exposure comparable to the thromboprophylactic 10 mg/day dose in adults
- Aspirin dose: 5 mg/kg/day, up to a maximum of 81 to 100 mg/day (per local practice)
OUTCOMES
- Primary efficacy outcome: Any thrombotic event, venous or arterial
- Safety outcomes: Major bleeding events (primary); nonmajor and trivial/minimal bleeding (secondary)
Not powered to test formal hypotheses for efficacy and safety due to the limited availability of the study population and the expected low event rates.2,3
BID = twice a day; eGFR = glomerular filtration rate; NSAIDs = nonsteroidal anti-inflammatory drugs.